In mid-2016, a new set of regulations concerning product compliance with European industry standards will be put in place for pharmaceutical and life science organizations. Identification of Medicinal Products (IDMP) is a framework of detailed descriptions of substances, composition and dosage forms, production procedures, and packaging. These IDMP norms require identification of all pharmaceutical products according to certain data standards, laid out by ISO (International Organization for Standardization). ISO has come up with five IDMP standards; these are aimed at accurately identifying medicinal products for human use, with a high degree of certainty.

With Europe being the first region to adopt Identification of Medicinal Products (IDMP) standards, by July 1st, 2016, time is running out for life science organizations to comply. It is anticipated that the U.S. Food and Drug Administration (FDA) and the Japanese Pharmaceutical and Medical Devices Agency (PMDA) will follow the European Medicines Agency’s (EMA) advance move. EMA is the first regulatory agency that requires life science organizations to comply with the ISO standards. In the case of noncompliance, organizations will face severe fines. Life science organizations therefore will need to quickly establish a data standardization process that is robust, reliable, and flexible enough to meet these varying regulatory demands.
The main challenges from the perspective of implementing compliance are:
- An enormously ambitious time schedule. Life science organizations are obliged to adhere to the requirements prior to the July 2016 deadline. But the final EMA implementation guidelines were only made available in late 2015. This leaves organizations with a time slot of less than a year to comply with the new standards.
- The need to collaborate between business units. The product master data required to satisfy the IDMP standards is present in a wide set of units and systems within life science organizations and their suppliers.
- A massively unstructured data pool. So far, most data has been submitted to regulators in heterogeneous formats such as pdf, doc and txt files. Details about substances such as the Summary of Product Characteristics (SmPC), Manufacturing Licenses, Chemistry, Manufacturing and Control (CMC) documents and others are present in a wide set of source systems within the organizations.
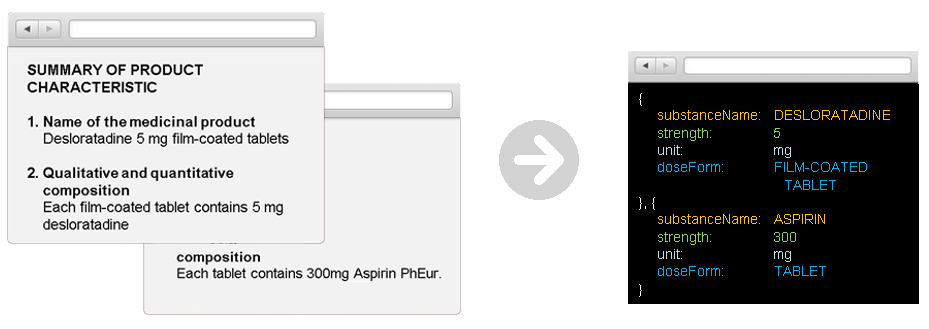
Regarding the last point, text mining technologies are crucial for overcoming the complexity and heterogeneity of unstructured data. Text mining refers to the process of deriving high-quality information from text, a process that is capable of quickly extracting relevant information from text sources and structuring heterogeneous data contained in various IDMP relevant sources, such as SmPC documents. Relevant information includes product names; ingredients; excipients; pharmaceutical dosage forms, strengths, and units; undesirable side effects; and much more. This information is mapped to standardized vocabulary as defined in the above-mentioned ISO standards and as shown in the following picture.
Let’s take undesirable effects as an example, to show the complexity of the task and the capabilities of advanced text mining solutions. Side effects are usually listed in section 4.8 “Undesirable Effects” of SmPC documents. They are mostly present in different table formats but can also be found in a text passage. Side effects will be coded/mapped to the MedDRA vocabulary. The information intended for extraction consists of multiple items from the table. For example, adverse events are usually listed in connection with its System Organ Class (SOC) and frequency. Text mining solutions must be able to extract such complex information and relations from tables and automatically map it to MedDRA codes.
If a text mining solution is able to fulfill these needs with high precision, then it will save pharmaceutical companies a lot of time and money. It creates a consistent and homogenized set of product master data, permitting significant analysis. This delivers new insights in areas including post-marketing surveillance, competitor analysis, supply chain, and sales/marketing. Expenses associated with managing, integrating, maintaining, and reconciling data across functions and sites are reduced.
Averbis offers solutions that enable organizations to comply with IDMP, within the given timeframe, and to leverage data assets. Please contact us to find out more.

